| Home | Scope | | | Types/Configs. | | | Schematics | | | Heat Balance | | | Heat Trans1 | | | Heat Trans2 | | | Process | | | Circulation | | | Other | | | Publications | | | Calculators |
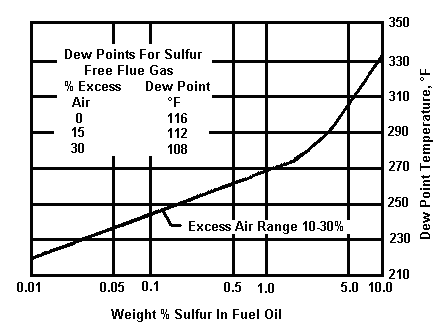
To improve the thermal efficiency of combustion equipment it is necessary to cool the flue gas to a low outlet temperature, to recover as much heat as possible. But the temperature must not be so low as to allow sulphur from the fuel to condense as sulphuric acid, resulting in a very corrosive flue gas.
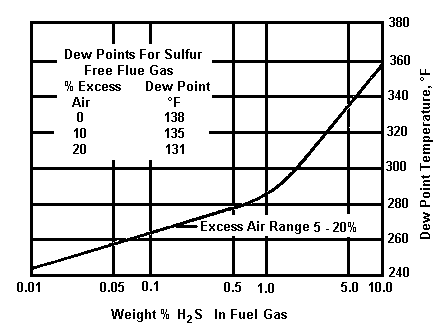
The two graphs below indicate the maximum flue gas dew point versus sulphur content in typical fuels.
Maximum flue gas dew point versus percent sulphur in typical oil fuels:
Heat Recovery Steam Generators (HRSGs), do not form nitrogen oxides or other contaminants. However, the supplementary burners associated with this equipment, or the source of the flue gasses, such Gas Turbines or FCC Units, do create the conditions for the formation of these undesirable pollutants.
Even though, NOx is not the product of combustion such as CO, CO2, and SO2, they are a side effect. When the fuel and air are mixed, as they are in the combustion process, the nitrogen, which is normally very stable, can react with other substances to form Nox. This reaction at higher temperatures causes the nitrogen to react with oxygen to form NO, and N2O referred to as NOx.
Even with the available control methods such as steam/water injection in Gas Turbines and staged burner combustion in other equipment, there is sometimes the need to further reduce the NOx in the gas stream to the HRSG. Because of the low temperatures normally involved, this is normally accomplished with a Selective Catalytic Reduction (SCR) technology.
SCR is the most effective and proven technology to reduce NOx emissions. With guaranteed reductions greater than 90%, SCR is adaptable to a wide range of temperature and service conditions. The SCR systems reduce NOx by optimally distributing ammonia over the catalyst bed, reducing NOx to harmless nitrogen and water.
SCR, however, is not without its problems. The first is that the excess ammonia from the process is exhausted into the air. Both NOx and ammonia are acutely toxic, both contribute to fine particle formation, acidifying deposition, eutrophication and enrichment of terrestrial soils; and both may be converted to nitrous oxide (N20).
Anhydrous ammonia (NH3) is the most economical reagent to use, however, the growing trend by end-users and operators is the use of aqueous ammonia (NH4OH). Aqueous ammonia is a safer reagent to transport, handle and store than anhydrous ammonia. Vaporization of the aqueous ammonia solution is required to reach the base ammonia yield needed for the reaction. Typically, this vaporization is accomplished by using a hot air source. Exhaust gas can be withdrawn from the, or ambient air can be heated.